Eureka, I would like to share my latest plating results with you. You might know that I am very interested in isolating any kind of wild yeasts from commercial sour beers. The most difficult task in this whole isolation process is to differentiate normal Saccharomyces cerevisiae colonies from other yeast species such as Brettanomyces.
Previous studies to develop new kind of agar media to detect Brettanomyces in wine samples showed bromocresol green to be a useful indicator to detect acid producing Brettanomyces strains [Rodrigues et al., 2001; Couto et al., 2005, EP 1185686 A1]. In this case, bromocresol green acts as a pH-indicator and turns yellow in the presence of acid which is produced by some Brettanomyces species. The authors further added cycloheximide to the media to prevent any growth of Saccharomyces. Concluding from the previously cited publications an addition of cycloheximide to agar media should already be enough to differentiate between Saccharomyces and Brettanomyces colonies by simply inhibiting the growth of Saccharomyces. Further antibiotics could be added to prevent the growth of bacteria.
Other studies showed that bromocresol green alone can be used to differentiate between the two yeasts in absence of antibiotics [Yakobson, 2010]. In addition, bromocresol can diffuse into yeast colonies and form green colonies due to the accumulation of the dye [Yakobson, 2010]. However, some Brettanomyces strains seem to be able to form white colonies again. This has been shown in other experiments as well [Rodriguez, 2012; BKYeast, 2012]. Yakobson mentions that the dye gets actively metabolized by Brettanomyces and hence the white colonies again. Unfortunately, I could not find any source investigating how exactly the dye get metabolized. Yakobson further mentions that some Saccharomyces strains can form white colonies as well which would make it even more difficult to differentiate between Saccharomyces and Brettanomyces.
The aim of this study was to screen different Saccharomyces strains for their ability to form white colonies on bromocresol green containing agar.
Material
- Sabouraud agar 4% glucose, Art. X932.1, Roth
- Bromocresol green sodium salt, Art. KK18.1, Roth
- Saccharomyces strains from Wyeast and White Labs
19 different Saccharomyces strains including one Saccharomyces mixture (WY3056) were plated on Sabouraud agar containing bromocresol green. Bromocresol green was added as aqueous, sterilized solution to the sterilized Sabouraud agar until the agar turned blue. The plates were incubated at room temperature at a dark place until colonies were visible. A control was included (no yeast streaked) to observe any color changes of the agar due to environmental effects (photo bleaching, oxidation, decay etc). The following yeast strains were used for this screen.
| Number |
Product name |
| WY1010 |
American Wheat |
| WY1056 |
American Ale |
| WY1084 |
Irish Ale |
| WY1728 |
Scottish Ale |
| WY1762 |
Belgian Abbey II |
| WY2112 |
California Lager |
| WY2278 |
Czech Lager |
| WY2487 |
Helle Bock |
| WY3056 |
Bavarian Wheat Blend |
| WY3068 |
Weihenstephan |
| WY3333 |
German Wheat |
| WY3522 |
Belgian Ardennes |
| WY3638 |
Bavarian Wheat |
| WY3711 |
French Saison |
| WY3726 |
Farmhouse Ale |
| WY3864 |
Canadian/Belgian Ale |
| WY3942 |
Belgian Wheat |
| WY3944 |
Belgian Wit |
| WLP002 |
English Ale |
| Control |
N/A |
Results Part 1
Colonies were visible after four days of incubation (Fig 1-5). The control showed no colony formation and the agar showed no color change. The colors of the agar were compared with the control.

Fig 1: Yeasts on bromocresol agar after four days. Left: WY1762, Top: WY3522, Right: WY3864; Bottom: Control
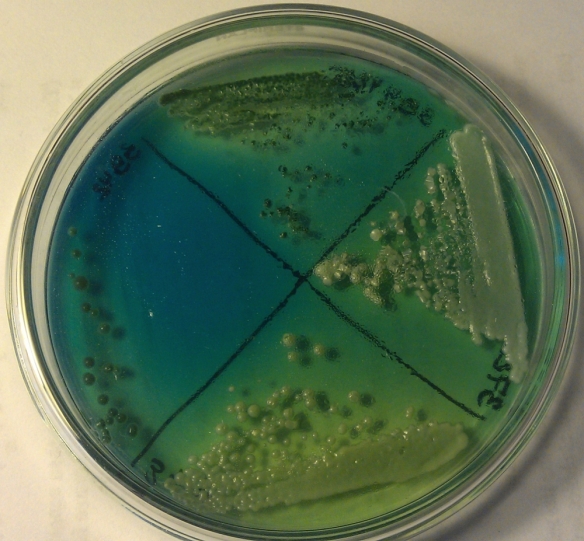
Fig 2: Yeasts on bromocresol agar after four days. Left: WY3942, Top: WY3944, Right: WY3726; Bottom: WY3711

Fig 3: Yeasts on bromocresol agar after four days. Left: WY2487, Top: WY2112, Right: WY2278; Bottom: WY1010

Fig 4: Yeasts on bromocresol agar after four days. Left: WY3638, Top: WY3068, Right: WY3333; Bottom: WY3056

Fig 5: Yeasts on bromocresol agar after four days. Left: WLP002, Top: WY1056, Right: WY1728; Bottom: WY1084
Some Saccharomyces strains were able to change the color of the agar from green-blue to yellow. Only two yeast strains, WY3333 German Wheat and WY3726 Farmhouse Ale, grew as white colonies on bromocresol green agar after four days. This already is proof that some strains indeed can grow as white colonies on bromocresol green. The plates were further incubated and after a total of twelve days, the color of the colonies were evaluated for a second time (Fig 6-10). Sorry for the bad quality of the pictures.

Fig 6: Yeasts on bromocresol agar after 12 days. Left: WY1762, Bottom: WY3522, Right: WY3864; Top: Control

Fig 7: Yeasts on bromocresol agar after 12 days. Left: WY3942, Top: WY3944, Right: WY3726; Bottom: WY3711

Fig 8: Yeasts on bromocresol agar after 12 days. Left: WY2487, Top: WY2112, Right: WY2278; Bottom: WY1010

Fig 9: Yeasts on bromocresol agar after 12 days. Left: WY3638, Top: WY3068, Right: WY3333; Bottom: WY3056

Fig 10: Yeasts on bromocresol agar after 12 days. Left: WLP002, Top: WY1056, Right: WY1728; Bottom: WY1084
One could observe that some of the colonies now have white edges and a green centre. All these colonies were still counted as green colonies.
This time less yeast strains turned the agar to a yellow color because the control agar lost a lot of its blue color. The global decrease of the blue color in the agar might originate from diffusion of acids secreted by yeasts that turned the agar yellow. Or due to the diffusion of the dye into the colonies. Further on to the white yeast colonies. WY3333 German Wheat and WY3726 Farmhouse Ale still grew in white colonies. In addition, WY3864 Canadian/Belgian Ale and WLP002 English Ale now grew as white colonies as well. One might expect further yeast strains to form white colonies with a prolonged incubation time because a lot of the colonies already have white edges and a remaining green centre.
After 17 days of incubation, the colonies looked as shown below (Fig 11).

Fig 11: Yeasts on bromocresol agar after 17 days. Left/bottom: WY2487, Left/Top: WY2112, Right/Top: WY2278; Right/Bottom: WY1010
A lot of yeast colonies now turned into white colonies as expected (Fig 12). The color was now evaluated by looking at the entire colonies visible for a particular strain. If more than 50% of the colonies were white, the yeast was counted as white. Like the WY2278 Czech Lager shown in Fig 11. On the other hand, all the other yeasts shown in Fig 11 were counted as green like the WY3711 French Saison in Fig 12.

Fig 12:WY3711 French Saison colonies after 17 days of incubation
After 17 days of incubation, only seven out of the 19 screened yeasts still had green colonies. All the other ones turned white in the meantime. To put it in numbers. After 4 days 2/19, after 12 days 4/19 and after 17 days of incubation 12/19 yeast strains formed white colonies (Fig 13). This clearly shows a time dependency.

Fig 13: Yeast screen results on heavily stained bromocresol green agar
Results Part 2
The blue color in the agar plates (Fig 1-5) was quite heavy and to test whether a lower concentration of bromocresol green in the agar leads to the same results as discussed above, a second experiment was conducting by streaking the exact same yeast strains on some Sabouraud agar containing bromocresol green. This time a lower concentration of bromocresol green was used.
Quantification of the color after three days of incubation (Fig 14-18):

Fig 14: Yeasts on bromocresol agar after three days. Left: WY3864, Top: Control, Right: WY1762; Bottom: WY3522

Fig 15: Yeasts on bromocresol agar after three days. Left: WY3942, Top: WY3944, Right: WY3726; Bottom: WY3711

Fig 16: Yeasts on bromocresol agar after three days. Left: WY2112, Top: WY2278, Right: WY1010; Bottom: WY2487

Fig 17: Yeasts on bromocresol agar after three days. Left: WY3638, Top: WY3068, Right: WY3333; Bottom: WY3056

Fig 18: Yeasts on bromocresol agar after three days. Left: WLP002, Top: WY1056, Right: WY1728; Bottom: WY1084
Yet again some colonies grew as white colonies and others grew as green ones (Fig 19). Comparing the results with the one concluded from the first experiment, WY3726 Farmhouse Ale, WLP002 English and WY3864 Canadian/Belgian Ale showed white colonies. In contradiction with the first experiment are the color morphologies of WY3333 German Wheat, WY1728 Scottish Ale and WY3711 French Saison. WY3333 grew as white colonies in the first experiment and as green ones in the second one. On the other hand, WY3711 and WY1728 grew as white colonies in the second experiment.

Fig 19: Closer look at Fig 15
The colours were again determined after further incubation. Agar plates shown after 12 days of incubation (Fig 20-24).

Fig 20: Yeasts on bromocresol agar after 12 days. Left: WY3864, Top: Control, Right: WY1762; Bottom: WY3522
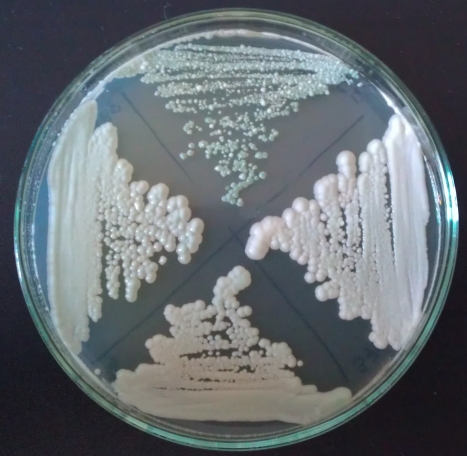
Fig 21: Yeasts on bromocresol agar after 12 days. Left: WY3942, Top: WY3944, Right: WY3726; Bottom: WY3711

Fig 22: Yeasts on bromocresol agar after 12 days. Left: WY2112, Top: WY2278, Right: WY1010; Bottom: WY2487
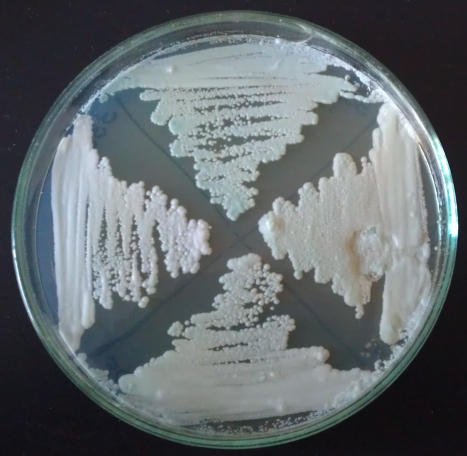
Fig 23: Yeasts on bromocresol agar after 12 days. Left: WY3638, Top: WY3068, Right: WY3333; Bottom: WY3056

Fig 24: Yeasts on bromocresol agar after 12 days. Left: WLP002, Top: WY1056, Right: WY1728; Bottom: WY1084
Twelve days of incubation and all the yeast strains have the same color like a few days ago. The plates were further incubated and a final color determination was conducted after 17 days (not shown).
The results of the second run are summarized in Fig 25. WY1010 American Wheat, WY1084 Irish Ale, WY1762 Belgian Abbey II, WY2112 California Lager, WY2278 Czech Lager, WY3068 Weihenstephan and WY3942 Belgian Wheat all had white colonies after 17 days (Fig 25). 13/19 yeast strains grew as white colonies after 17 days of incubation (Fig 25).
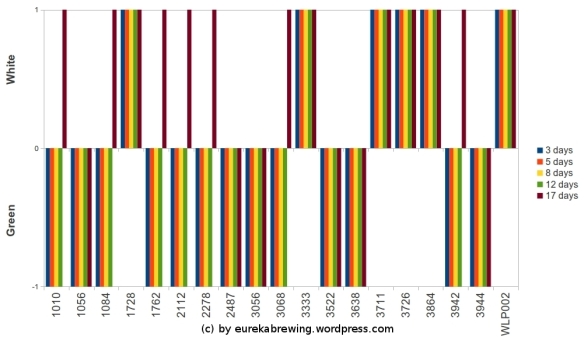
Fig 25: Yeast screen results on light-stained bromocresol green agar
Discussion
Comparing the two experiments, some strains such as WY1010 American Wheat, WY1728 Scottish Ale, WY2112 California Lager, WY3711 French Saison and WY3942 Belgian Wheat only grew in white colonies after 17 days on the light stained agar media and not the heavy stained one (Fig 26). WY3056 Bavarian Wheat Blend, WY3522 Belgian Ardennes and WY3638 Bavarian Wheat grew as white colonies on heavily stained agar but as green ones on lightly stained agar media (Fig 26). This might be an indicator that the bromocresol green concentration might influence the color change as well.

Fig 26: Differences between the two experiments
As a general trend, the different yeast strains seem to form white colonies after further incubation. However, two strains (WY3333 German Wheat and WY3726 Farmhouse Ale) grew on heavily stained agar as white colonies from very early on (Fig 13) and four additional ones on lightly stained agar (Fig 25). Yakobson states on his website that Wit yeasts can metabolize bromocresol green (http://www.brettanomycesproject.com/2009/03/wln-agar-medium/). In this screen the Wit strain from Wyeast (WY3944) did not grew as white colonies in both experiments (Fig 13, 25). Not even after 17 days of incubation.
Some words about the color of the agar media. Fig 1 to 5 are nice examples to show that the color of the bromocresol containing media changes its color from green to yellow. In both experiments, the color of the control agar turned to a yellow color as well. The plates were stored at a dark place to prevent any influence of light (photobleaching effects). The change in color might be due to secretion of acids (bromocresol changes color at lower pH to yellow), due to a take-up of the dye by the yeast cells like stated by Yakobson in case of Brettanomyces. Another possibility might be the stability of bromocresol green itself. If one imagines bromocresol green to be a relatively unstable molecule, the loss of the green color might be due to the depletion of the dye. Yakobson further mentions that Brettanomyces can even metabolize the dye and therefore grow as white colonies. All the cells not able to metabolize the dye remain as green colonies. Unfortunately, I could not find any evidence for this statement showing that Brettanomyces really metabolize the dye. Nor any evidence that Saccharomyces can do it. Maybe the cytoplasm of Brettanomyces cells have a lower pH and therefore turn the dye from green to yellow. There might even be some truth about this hypothesis since some Brettanomyces strains are known to secrete acetic acid under aerobic conditions. It is therefore not clear to me why/how the colonies turn from green to yellow.
I would like to discuss bromocresol green as a useful tool to differentiate between Brettanomyces and Saccharomyces. Although I did not show any Brettanomyces colonies here, the bromocresol screen strongly suggests that some Saccharomyces strains can grow as white colonies on bromocresol green containing agar media. This makes a differentiation already a bit harder. In addition, a majority of Saccharomyces yeast strains appear as white colonies after a longer incubation period. BKYeast came to the conclusion that differentiation based on bromocresol green might only be possible in a short time frame in mixed cultures (Saccharomyces and Brettanomyces grow on the same plate). The results from these experiments show that even in pure cultures, and in absence of Brettanomyces, a lot of the Saccharomyces strains tested turned from green to white within a short period of time. All these results strongly suggest that any differentiation solely based on bromocresol green might only be useful in a short period of time.
Summary
Bromocresol screen is a widely used differentiating dye to differentiate between Saccharomyces and Brettanomyces. Brettanomyces known for their capability to grow as white colonies while Saccharomyces grow as white ones. It has been reported that some Saccharomyces strains grow as white colonies as well and therefore making a differentiation more difficult [Yakobson, 2010]. Screening different Saccharomyces cerevisiae strains on bromocresol green containing Sabouraud agar revealed some strains capable of growing as white colonies from the very beginning where the majority of yeast strains grew as green ones. Therefore showing that indeed some yeast strains can grow as white colonies. After further incubation, the majority of the yeast strains turned from green to white coloured colonies. There seems to be a general trend for Saccharomyces cerevisiae strains to form white colonies after extended incubation times. However the reason for this observation is not clear at this point as well as the mechanism leading to the observed change in color. It can’t be excluded that different sources like instability of bromocresol green itself or any environmental factor lets the colonies turn from green to yellow.
Due to these observations, bromocresol green as a tool to differentiate between Brettanomyces (known to grow as white colonies) and Saccharomyces might only work within a small time frame. This has been previously observed by BKYeast as well.
Outlook
Brettanomyces bromocresol green screen similar to the one shown here for Saccharomyces. In addition, try to grow Brettanomyces anaerobically to test whether the colonies grow as white or green ones (acid theory mentioned in the discussion).
References:
- Couto J.A., Barbosa A. and Hogg T. (2005) A simple cultural method for the presumptive detection of the yeasts Brettanomyces/Dekkera in wines. Letters in Applied Microbiology, 41, 505-510
- BKYeast (2012) http://bkyeast.wordpress.com/2012/02/19/selective-media-part-i/
- EP 1185686 A1, Culture medium for detection of Dekkera and Brettanomyces, http://www.google.com/patents/EP1185686A1?cl=en
- Rodrigues N., Goncalves G., Pereira-da-Silva S., Malfeito-Ferreira M. and Loureiro V. (2001) Development and use of a new medium to detect yeasts
of the genera Dekkera/Brettanomyces. Journal of Applied Microbiology, 90, 1-12
- Rodriguez J. (2012) Brooklyn Brewery Samples on New Wild Yeast Media, sciencebrewer.com
- Yakobson C. (2010) Brettanomyces project, http://www.brettanomycesproject.com/2009/03/wln-agar-medium/
I am open to any discussions and feedback concerning this experiment. Thank you for reading.
 Official tasting number 1, (08/30/2011), two months in bottle
Official tasting number 1, (08/30/2011), two months in bottle

































