Eureka, today I proceed with the series about yeast banking at home (or in a lab). The introduction (previous post) about yeast banking can be found here. Lets begin with technique number one, yeast banking with agar plates.
Description of the technique
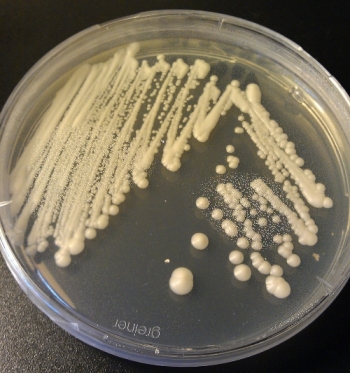
Agar plates are basically petri dishes (Fig 1) which are filled with a gelatinous media called agar media. For that you mix yourself a agar media, heat it up and sterilize it and then pour the media in the petri dishes. As the media cools down, it gets hard and dry.
The agar media usually consists of nutrients for the microorganisms to grow and agar agar (or just agar) which makes the whole agar media gelatinous. Agar is very similar to gelatin which is used in cooking. More about the agar media later on.
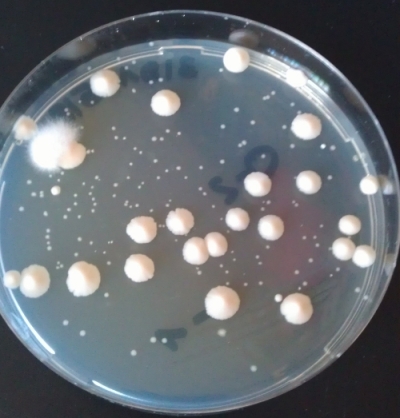
Agar plates are very widely used in biology labs to cultivate microorganisms such as bacteria and yeasts. For that you basically distribute a liquid sample (also known as plate or streak) with your suspended microorganisms on a agar plate and wait for colonies to arise (Fig 2). The colonies are the spots you can observe. Colonies arise from one single cell (in theory) and form a colony due to the increasing amount of cells at the same spot and become visible. Meaning, a colony are a lot of cells originating from a single cell. If you plate a defined volume of your sample on a plate you can even determine the amount of microorganisms you have in your sample. Agar plates are therefore a very basic technique in microbiology.
Not only can you cultivate the microorganisms but differ between some of them if you choose the right kind of agar. Meaning, some bacteria/yeast can grow on one kind of agar and others can’t. The trick here is to use the right kind of agar. You can even add further substances such as antibiotics to avoid bacterial growth on the plate, add dyes to watch for colored colonies, add pH-indicators to watch for colonies producing acids or add further substances to avoid yeast growth… the possibilities are huge.
Lets go back to the yeast banking. Agar plates can be used to bank yeast as well (what a surprise). The easiest agar for homebrewers is malt agar which is based on dry malt extract and agar agar to get the gelatinous consistency. Recipes can be found on BKYeasts blog as well as other agar media recipes. Malt agar is not the only way to go. I use Sabouraud agar for example which is a special agar to cultivate yeasts. More about Sabouraud and my agar plating technique can be found here in a very scientific post. In the end, it does not mater which kind of agar you use as long as yeasts can grow on them. The technique and material stay the same. For beginners, malt extract agar is probably the best way to go since dried malt extract is available at homebrew suppliers. A recipe for malt agar can be found on BKYeasts blog (look for malt extract/ wort agar). Don’t bother about the pH. You basically need dried malt extract, agar agar and a way to get the whole media sterile. Unfortunately, I can’t tell you about any sources for agar. In my case, agar is available at supermarkets because agar is used in baking as well.
Material
For yeast banking with agar plates you need:
– Petri dishes (available in glass or plastic)
– Source to get the agar media sterile (for example a pressure cooker)
– inoculating loop (DIY works very well)
– Source to flame the inoculating loop
– Agar (aka agar agar)
– Dried malt extract (aka DME)
Concerning the Petri dishes. Petri dishes are available at Amazon for example. Either go for glass or plastic dishes.
- Glass petri dishes can be heat sterilized in a pressure cooker or oven. Are more expensive than plastic ones. Can be re-used.
- Plastic petri dishes are normally just used once. They usually melt during a heat sterilization step… Plastic dishes are available as sterile and non-sterile ones. Some dishes have vents to allow gas exchange. In my opinion vents are great. However not necessary.
Next thing to remember is buying petri dishes with a cover. Concerning the diameter. There are different sizes of petri dishes available. Common diameter for dishes is about 94 mm. However, you can certainly buy bigger or smaller ones if you want. I use 94 mm diameter non-sterile plastic dishes because I do a lot of platings and do not sterilize the plates before use. Don’t had a problem with contaminations so far. If you plan on doing a lot of platings, get yourself some plastic dishes.
Source to get the agar media sterile. This is a very important step to consider. Your media has to be sterile and heated up to a boil. This is important since the agar will otherwise not be dissolved properly and not gelatinize the agar media. A pressure cooker would be the best thing to have since the pressure will prevent any boil-overs.
Inoculating loops can be bought from Amazon as well or make one yourself. It is basically a metal loop… which has to be heated up before you distribute the yeast on the plate. This is done by a flame. Gas burners, ethanol burners, candles or any other flame will do the job. For further information please have a look at the Braukaiser’s post about how to make plates and slants.
Preparation
1. Make sure you have all the necessary equipment ready
2. Make some plates according to the Braukaiser’s post. I do not want to explain all the necessary steps in this blog since others have covered this topic already.
Just remember, the Braukaiser uses glass petri dishes which can be sterilized in a pressure cooker. If you work with plastic dishes you need to sterilize the media first since the plates will probably melt during the sterilization process. On the other hand, if you sterilize your glass petri dishes with the media the plates could be very moist afterwards. And water on the plate can smear your yeast colonies afterwards… You will not get nice colonies.
3. After the plates cooled down, they are ready to be streaked. Once again, one technique is described on the Braukaiser’s blog on inocculating plates and slants. For the visual people, have a look at the following YouTube video on How to Streak a plate.
4. Leave the plate(s) at a warm plate for some days until colonies appear. If you only have nice colonies such as the one shown in Fig 2, you basically mastered the agar plating technique already. Well done!
Bank the yeast
After colonies appeared, seal the plate with a tape and the plate is ready for storage. The tape prevents the plate to dry out and the introduction of any contaminations. However, don’t be too sure about the tape. Even sealed plates can get a contamination at some point.
Storage
Store the plate in a refrigerator at approximately 6°C (43°F). Avoid temperatures around 0°C (32°F) or below. You can even store the plates at warmer temperatures. However, in this case a lot of maintenance work will be necessary. At warmer temperatures the yeast still grow relatively fast and will sooner or later form very big colonies. Maybe even cover the whole agar plate. In this case, you need to pick a yeast colony and plate it on another plate (also known as re-streaking or re-plating). Incubate the plate again until colonies appear. Then pick another colony and plate it on another plate… You see where this is going. At cooler temperatures above freezing temperature, the yeasts will grow relatively slow. A re-streaking will be necessary at some point as well. However, the time between theses platings will be much longer compared to plates stored at ambient temperatures.
Reanimation
This is rather easy. Just pick a colony and make a small starter. More about this process in more detail again on Braukaiser’s blog about growing yeasts from a plate.
My experiences with this method
What are the advantages/disadvantages for this method as a banking tool?
| Advantage | Disadvantage |
| Rather easy method | Plates needs storage space |
| Not a lot of equipment necessary | Contaminations happen |
| Contaminations can be visible | Lot of maintenance work necessary |
| Agar plates can be used for other projects | Not a long-term storage method |
Let me talk about the advantages first. Compared to some other techniques in yeast banking this one is rather easy to do and you do not need sophisticated equipment. In addition, contaminations in your yeast can be visible very easy. This can be seen if any other colonies (different look) appear on the plate. If there are only one kind of colonies on the plate, the chance is high your yeast is still pure. However, a contaminations does not have to be visible… Some are not visible on the plates. Therefore, contaminations can be visible. At last, you can use the agar plates for other projects such as isolating brewer’s yeast or wild yeasts from commercial beers.
Disadvantages of the agar plates yeast banking method. If you have a lot of strains to bank, keeping them all on agar plates will use a lot of storage space. And a lot of maintenance work since you have to re-plate them periodically. And every re-streaking is an opportunity for a contamination to sneak in. As already mentioned, even a taped plate can get infected at some point. For all these reasons, banking yeasts on agar plates is not a long-term storage method in my opinion.
You might ask yourself why I wrote a post about agar plates as a banking tool after all. In my opinion, if you only use limited yeast strains (lets say three different ones) banking them on agar plates might work really well. Just pick a colony from a agar plate, make a starter and re-streak another plate with the starter’s liquid. With this method you do not only have fresh yeast on a plate but the opportunity to check for any contaminations in the starter as well.
If you do want to get into yeast stuff yourself, agar plates are a very basic technique. Even if you start your own yeast library with a different technique such as banking them in sterile solutions, slants or freeze them, agar plates still can be a useful tool to cultivate your yeasts. However, you can use the other methods and not use any agar plates as well.
At the end, let me write about contaminations. Have a look at Fig 2 again. This is how a pure, healthy yeast culture should look like on an agar plate. Now have a look at the following pictures:
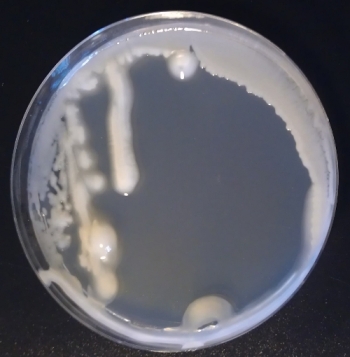
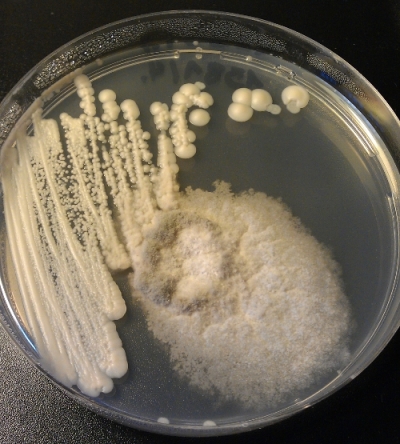
This is how a bacteria infection looks like (Fig 4). Can’t tell what kind of bacteria it was. But it was a bacteria (checked with microscope).
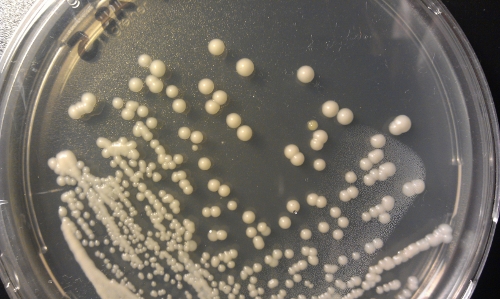
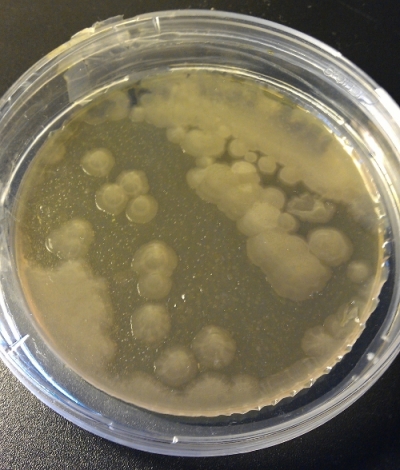
The next one is quite tricky (Fig 5). There is one colony which looks different… Found it? Will solve the puzzle later on… I have no idea what this was. Haven’t checked it out.
Next about molds: The white fluffy thing in the left upper corner (Fig 6).
Another mold (Fig 7). I do have a lot of mold contaminations. Maybe 80% of the contaminations I have are molds…
The last picture is no contamination. This is how a dry plate looks like (Fig 8). Chance is little to retrieve your yeast from such a plate… And the contamination colony in Fig 5 is the yellowish one on the right upper corner. Finding such odd-looking colonies can be hard. And if such contaminations appear you either get rid of the yeast or try to get rid of the contamination by re-streaking a colony on a new plate and hope for the best.
Unfortunately for me, Lactobacillus does not grow on Sabouraud agar. Lactobacillus is a very common beer spoilage bacteria. Another one is Acetobacter. If there is Lactobacillus in one of my yeast sample, I could not tell by just looking at the agar plate since no Lactobacillus colonies arise. Maybe someone out there has any experience with Lactobacillus on malt agar. I want to close this huge post with a thing to remember. Keep in mind, you just see the microorganisms capable of growing on the agar media. Any microorganisms not able to grow on the agar media will stay hidden… Stay tuned! The next post will be about banking yeast in sterile solutions.