Eureka, its time for another yeast basics post. This post is all about yeast viability. Viability describes how many living cells there are in a cell population. The higher the viability the more living cells you have. Lets apply this definition to homebrewing. A high viability is very important in brewing since only living yeast cells can ferment wort. Pitching the right amount of yeast cells is important. However, viability is important to consider in the pitching rate calculations as well. Pitching a yeast with a low viability is basically the same as underpitching. Not only is it important to pitch the right amount of yeast but having viable yeast cells as well. Let me walk you through some of the most important facts concerning yeast viability and homebrewing.
How do you determine the yeast viability?
I would like to show you one example how you can determine the yeast viability by use of a microscope. If you do not have a microscope or are not interested in how it is done, please skip this chapter.
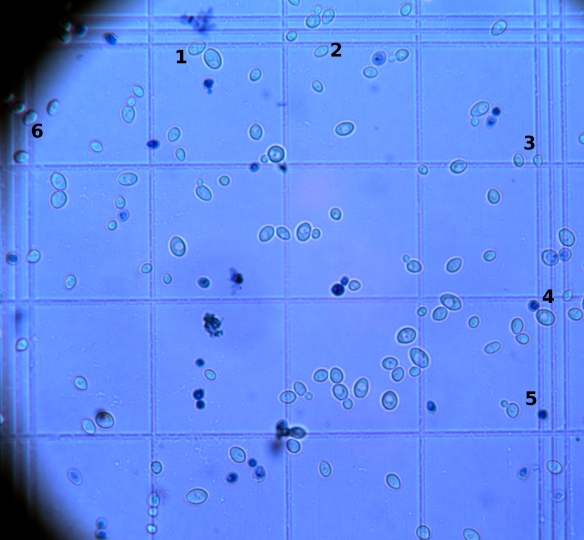
The easiest way to determine the viability is to stain yeast cells with methylene blue and count the yeast cells as described in a previous post about yeast counting. Methylene blue stains the dead yeast cells which then appear as dark blue cells. This could look like shown in Fig 1. The dark blue cells are dead, the other ones still alive.
To perform a viability count, mix equal parts of your yeast solution with a 0.1% (w/v) methylene blue solution. Mix well and let it react for one minute then count the cells by use of a counting chamber. Concerning the methylene blue solution. Mix 0.1 g of methylene blue and dissolve it in 100 mL of distilled water.
First, the cell counting is done the same way as you would do it without the methylene blue staining. The only difference, you count the dead cells in parallel in the different squares of the counting chamber. If you like further information about yeast counting, please have a look at a previous post of mine concerning yeast counting. Now, count all the yeast cells including the dead (dark blue) ones. Lets say you counted 105 cells in total. Second, you count all the dead ones. Lets assume you counted 15. Next, you calculate the viability:
Viability [%] = (Total counted cells – total counted dead cells) / total counted cells x 100
In our example, you get a viability of 86%. Meaning that 86% of the yeast cells are still alive. 14% are dead. For further information about the methylene blue staining please consult Hemocytometer.org and the book called Yeast: The Practical Guide to Beer Fermentation” written by C. White and J. Zainasheff.
Include viability in the pitching rate
Assume you want to ferment a 20 L (5.3 gal) ale batch with a gravity of 12°P (OG 1.048). In this case you need to pitch: 20000 mL x 12°P x 0.75E6 cells mL-1 °P-1 = 180E9 yeast cells. You can calculate the pitching rate with a calculator as well if you want. Lets assume you made a 1 L starter, diluted the sample by 1:100 and counted a total cell concentration of 1.3E9 cells per mL (including the dead yeast cells). To get the appropriate amount of yeast for pitching you need 138 mL of the starter (180E9 cells divided by 1.3E9 cells mL-1 = 138 mL). We also determined the viability in parallel to be 86%. In this case you would need around 160 mL of the yeast starter to have the right amount of yeasts (138 mL divided by 0.86 = 160 mL). You can easily see, the lower the viability is the more volume of the yeast starter is necessary.
How to determine the viability without counting yeast cells?
I assume most of the readers here do not have a microscope, therefore have no counting chamber and no methylene blue. How could you determine the viability without a microscope? As far as I know, there is no other way to do it. Well there are ways but these methods are even more expensive than a simple microscope, a counting chamber and methylene blue. The easiest way to do it is using assumptions. A few things to get good assumptions:
1. Using commercial liquid yeasts: As far as I know there is a production date on every liquid yeast package you buy from the main two yeast suppliers (Wyeast, White Labs). It is therefore possible to estimate the yeast viability based on the manufacturing date if the yeast is stored in a refrigerator:
Every day, you lose viability (Fig 1). Storing the yeast for a month, you already lost about 20% of the living yeast cells! Another month and you already lost nearly half of the living yeast cells! You can calculate the viability with J. Zainasheff’s (aka mrmalty) yeast calculator and the date printed on the yeast package. This works if you pitch the yeast from the package/vial directly. But please avoid pitching any yeast with a viability less than 90-80%. I recommend to make a yeast starter if the yeast sample has a low viability.
Please keep in mind, we are speaking about assumptions here. I am sure the viability decrease is dependent on the yeast strain, storage conditions, storage temperature, pH, oxygen level etc. etc. etc. However, this does not change the fact that yeast viability only decreases over time and therefore not recommendable to store yeast for longer than a couple of months.
2. Using yeast from a starter: If you do a yeast starter, only the viable cells will divide. The dead ones remain and decrease the viability as they are still present in the starter. If you do a starter from a Wyeast Activator package being 40 days old, the viability of the yeast is around 70% (Fig 1). Meaning, only 70% of the yeast cells are still alive. If you now do a 1 L starter with this yeast, the 30% dead cells will remain. The maximal viability you can obtain with this 1 L starter-step will be around 80% (Fig 2).
However, after the 1 L starter, you have approximately 125E9 viable yeast cells in the starter (Fig 3). A Wyeast package originally contains about 100E9 cells. Meaning, if you store your Wyeast package for 40 days, the viability is at 70% (Fig 1). From the original 100E9 cells, only 70E9 will be still viable. Then do a 1L yeast starter and your viability afterwards will be around 80% (Fig 2) and you have 125E9 viable yeast cells in total (Fig 3). Let me explain how the chart in Fig 3 must be read to get to the 125E9 cells. As mentioned a Wyeast Activator package has roughly 100E9 viable yeast cells at the beginning. Then store it for 40 days and do a 1 L yeast starter. Use Fig 3 to get from the 40 days to the 125% (or the equation). For the equation, use 40 days as x to get the 125% (as y). This means, from the original 100E9 viable cells you now have an increase of 125%, which is equal to 125E9 viable yeast cells in total.
Please notice, Fig 3 only works for Wyeast Activator packages and White Labs vial with an original yeast amount of 100E9 cells and a 1 L starter size. However, the equation from Fig 3 does not work for other yeast samples and other starter sizes. For these cases, use the calculator mentioned below.

Fig 3: Increase of (viable) yeast cells from Wyeast Activator package/ White Labs vial after a 1 L starter step vs. yeast age
Things to remember: The viability of yeast decreases over time (Fig 1). On the other hand, you cannot reach a 100% viability of an old yeast sample since the dead cells remain (Fig 2). The older a yeast sample is, the harder it is to increase the viability. And to estimate the viable yeast cells of a yeast sample after a 1 L yeast starter, use Fig 3.
Lets make a final example to show you how to use the charts:
Assume you want to ferment a 20 L (5.3 gal) ale batch with a gravity of 8.8°P (1.035). For this batch you need to pitch 132E9 cells. Assume you have a Wyeast package or White Labs vial being 30 days old. Now, use Fig 1 to determine the viability which is 76%. The Wyeast Activator/White Labs vial therefore only has 76E9 viable cells left. So not enough for a direct pitch. You therefore need to do a 1 L yeast starter and you will have 132E9 yeast cells at the end (Fig 3). That is just enough yeast to pitch. You therefore can pitch the yeast from the whole 1 L starter and have the right amount of yeast.
Because these charts only work for 1 L starters, use the yeast calculator from J. Zainasheff (mrmalty.com) for any other batch sizes, starter sizes etc. In this particular calculator all the assumptions and such are already implemented. I really like programs where you can fill in numbers and you get a result (such as the yeast calculator). However, now you know some of the equations behind the yeast calculator and how it maybe work. I am not related to J. Zainasheff or have any financial benefits from referring to his site. I do so because I use his calculator for some time now. I wrote my own calculator based on my own assumptions a few years back and luckily for me, the results from my calculator are very close to the one from J. Zainasheff. So no need for me to improve my calculator any further.
I like to emphasize again, all the charts shown in this post are based on assumptions. I would not be surprised if someone comparing the calculated viability done with the graphs above and a real cell count encounters differences. In addition, I would not be surprised either if differences between yeast strains exist as well. Even a cell count done with a counting device is not 100% correct. Luckily for us, brewing beer is not like rocket science and does not have to be very precise. Cheers on that!



Yet another great post Sam. I for one appreciate the effort that goes behind these type of posts.
Your are most welcome. Cheers
Sam
Very nice, Sam. I’ve been looking into Lactobacillus viability over time, since you mentioned it a while back, and to be honest I have NO CLUE what’s going on. It’ll have to wait some more time, but it looks like they’re quite viable even after over a year in the fridge…
I can’t remember where I originally picked that up. I just prepared some Lactobacillus vials for cryo storage anyway 😉 Thanks for sharing your experience with the Lactobacillus storage. An empirical prove that it is possible to cryo store Lactobacillus… so long literature knowledge 😉
Cheers, Sam
Yes, great article again. I really like this series–I’m in the process of converting my yeast storage from slants to isotonic NaCl solution thanks to the earlier ones.
Surely the most frustrating aspect of yeast ranching so far is having to guess the pitch rate based on volume. Using rough estimates intended for washed yeast slurry (Wyeast’s method and MrMalty) has worked OK, but I have had problems. I had my first experience with noticeable diacetyl a few months ago with a beer I made with ranched yeast. It took forever to take off due to underpitching (the yeast was active and fresh from the stir plate, but it wasn’t enough), and then once it took off half of the yeast ended up in the blow-off tube. Since then I’ve been strongly considering a cheap microscope for cell counts.
Hi there, thanks for commenting. I hope your conversion to the isotonic NaCl solutions works as well as it did for me.
Indeed the determination of cell concentrations is not as straight forward as one would think. In my opinion, the most reliable way to go is doing a cell count with a counting chamber and a microscope. You don’t even have to worry about possible differences between top- or bottom fermenting yeasts or even yeast strains. Include a viability stain with methylene blue and you are on a professional level already.
I am sorry to hear about your experience with your ranched yeast. However, I would not blame the yeast ranching rather than the cell estimation. Your ranched yeast was probably fine but the cell concentration was maybe too low.
I don’t have a lot of experience with diacetyl but I would expect the diacetyl to decrease if you still have some yeast in the bottle, keg etc. and store the beer at a relatively warm temperature. Maybe a longer storage at warmer temperatures would help to decrease the diacetly?
Cheers, Sam
Yes, sorry, that is what I meant. The yeast is probably fine, but my cell count estimate based on sediment volume totally failed me that time. The diacetyl did eventually reduce in the bottles, but it was still pretty noticeable.
I’ll have to try again with the same or similar recipe but with a little more care on the pitch rate to see if I get the same result.
Pingback: Yeast Viability Over Time - To Brew a Beer
Pingback: Yeast Viability Over Time | My Blog